What is the Difference Between Dermatological Test and In Vitro Skin Irritation OECD 439 Test?
What is the Difference Between Dermatological Test and In Vitro Skin Irritation OECD 439 Test?
Contents
What is the Difference Between Dermatological Test and In Vitro Skin Irritation OECD 439 Test?
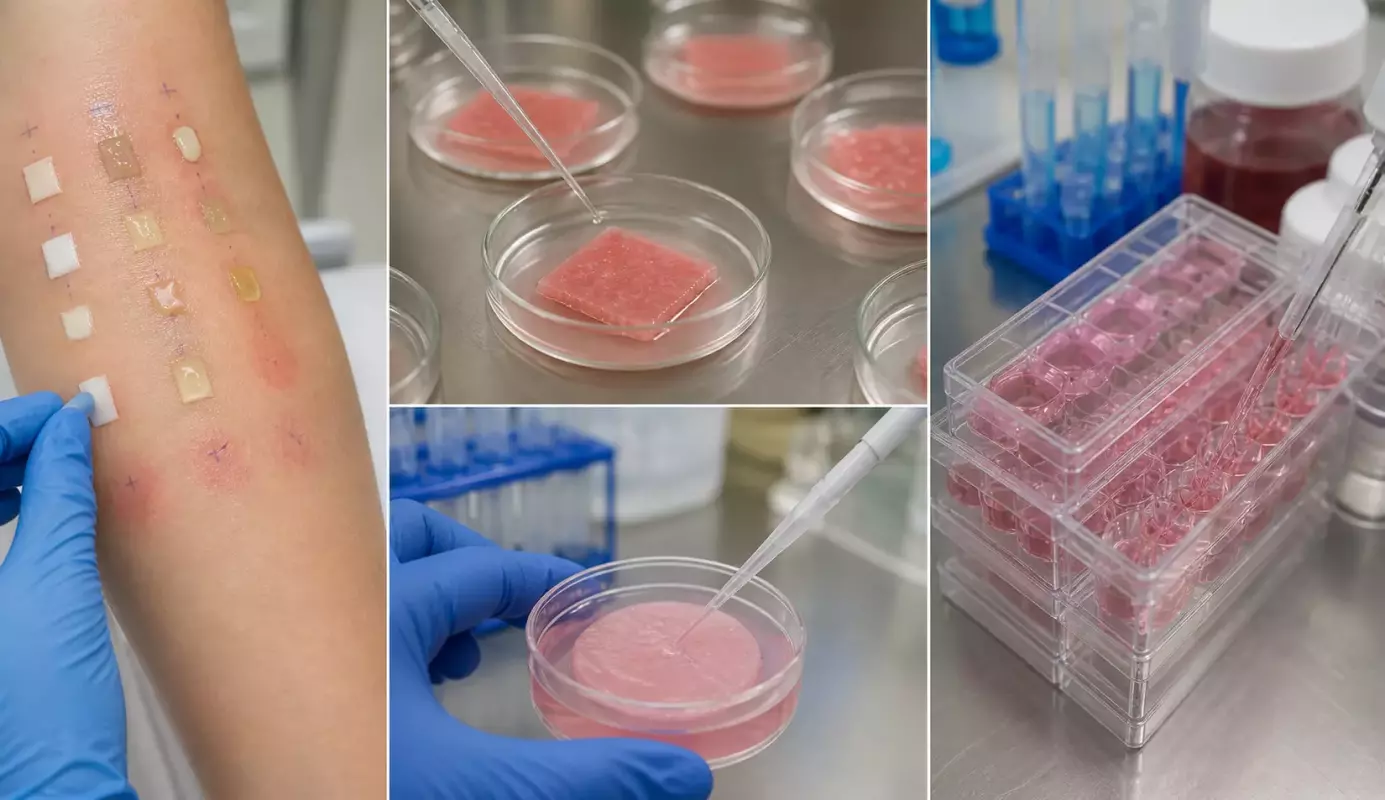
What is the Difference Between Dermatological Test and In Vitro Skin Irritation OECD 439 Test? is a key question in cosmetic safety evaluation. The difference lies in methodology, purpose, and application.
Dermatological Test
- In vivo testing on human volunteers
- Clinical observation by dermatologists
- Used for tolerance and marketing claims
OECD 439 In Vitro Skin Irritation Test
- Uses reconstructed human epidermis (RhE)
- Measures cell viability via MTT assay
- Classifies irritation potential
Standard: OECD 439
Key Differences
| Criteria | Dermatological Test | OECD 439 |
|---|---|---|
| Type | In vivo | In vitro |
| Model | Human skin | Artificial skin |
| Output | Clinical observation | Cell viability |
| Use | Marketing claims | Hazard classification |
Conclusion
What is the Difference Between Dermatological Test and In Vitro Skin Irritation OECD 439 Test?
They are complementary methods: in vitro testing ensures safety screening, while dermatological testing confirms real-life skin compatibility.

